Blood Transfusion Guidelines Updated
In an effort to improve patient safety and quality of care, BWH this summer updated its guidelines for transfusions of blood products following an intense review and analysis of the medical literature by a multi-disciplinary physician task force.
“With modifications to thresholds for transfusion of red blood cells, platelets and plasma, our hope is that BWH will see an overall reduction in the number of patients who receive transfusions and are consequently exposed to the risks associated with blood products,” said Richard Kaufman, MD, medical director of the BWH Adult Transfusion Service and an assistant professor of Pathology, who served on the 13-member task force.
In addition to improving quality and safety, it is hoped that the updated transfusion guidelines will help preserve the limited supply of blood products, especially during periods of low supply in the summer and around holidays.
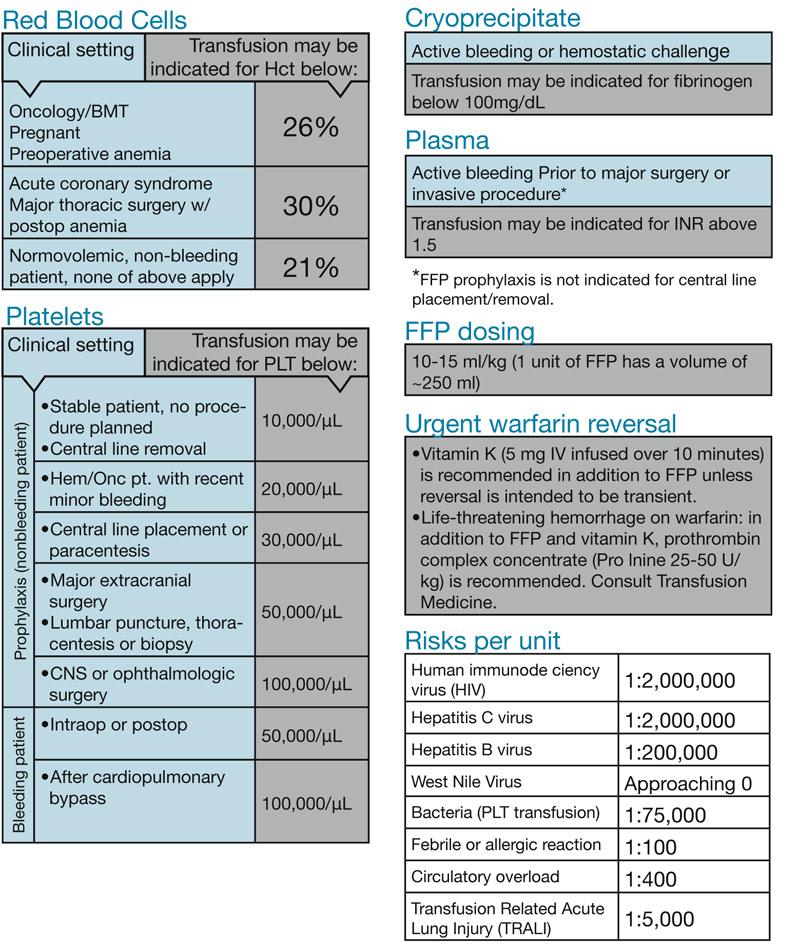
One major change for BWH clinicians comes with orders for platelet transfusions. The recommended platelet count that would trigger a transfusion prior to a central line placement is below 30,000/µL and, for central line removal, below 10,000/µL.
For most categories of patients, a red blood cell transfusion may be indicated if the hematocrit is 21 percent or below. For bone marrow transplant patients, pregnant patients or patients with preoperative anemia, transfusion may be indicated if the hematocrit is below 26 percent. For patients with an acute coronary syndrome, red blood cell transfusion may be indicated for a hematocrit below 30 percent.
For patients on warfarin with a major bleed, the task force recommends using vitamin K intravenously in addition to fresh frozen plasma transfusion. Certain patients may additionally benefit from receiving a prothrombin complex concentrate (PCC) infusion. This is a new standardized approach for BWH.
The physician task force included representatives from Hematology, Bone Marrow Transplantation, Thoracic Surgery, Cardiac Surgery, Anesthesiology, MICU, SICU, Medicine, Transfusion Medicine and the hospital administration. Following six months of review and development, the guidelines were approved by the BWH/DFCI Biological Therapies Committee.
The new guidelines are available on Partners Handbook, and a pocket card with specific details is available through the Blood Bank. Direct questions to the Blood Bank at 617-732-7290 or the Transfusion Medicine physician on call, beeper no. 35110.
Click image to enlarge